In order to replicate ecosystem and chemical origin of life, Stanley Miller, under the supervision of Harold Urey, performed the breakthrough Urey-Miller experiment in 1952. The experiment initiated more than 20 major molecules that form the integral part of life. A team of researchers at Stanford believes that they can do one-step better.
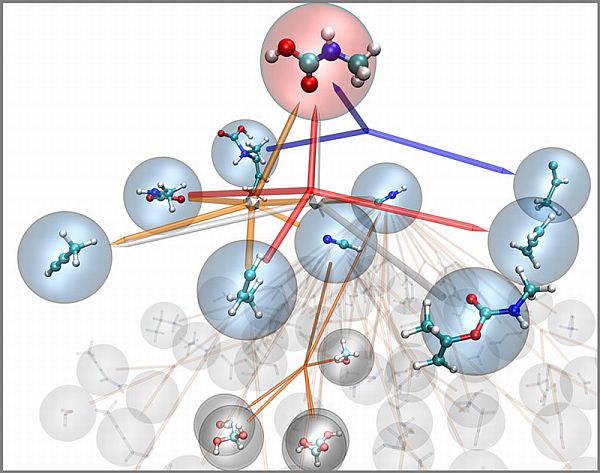
Nanoreactor
The team has developed a computer model that has not only the potential of determining the products formed but also the chemical reactions involved that could have yielded to one or more by-products. Researchers have named it the nanoreactor. Researchers claim that the model can surface the chemical reactions and hence envision that the discovery might help in enhancing fuel combustion efficiency.
Virtual chemistry lab
Being a part of the digital arena, the nanoreactor acts like a Chemistry lab, of course in a virtual world. For instance, by entering structures of specific chemicals into the simulator and setting the environmental conditions, that includes temperature and pressure, the algorithms starts solving the mechanical problems at quantum level. Movement of every electron within the molecules is recorded step-by-step along with the mechanisms involved.
The simulator surfaces all the important reactions that are going on with a single click of a button. The model is based on hybrid approach involving physics and machine learning to discern all the possible chemical reactions that might happen including the mechanisms that might otherwise have not witnessed before, added Todd Martinez, senior lead and Professor of Chemistry at Stanford.
Reaction network
Conventional approach involves jotting down single electrons movements, systematically, which is not only cumbersome but extremely complex as well. Manually reaching to the possible outcomes too constricts the limitations. However, running the algorithms on a computer is faster and reliable method as the simulation can render 100 to 200 atoms simultaneously and deliver results within couple of hours.
Improving energy production
Another major advantage of using the nanoreactor is to note the number of reactions involved in the real time, which however is very difficult to see through the combustion. Once there is an understanding of all the reactions, one can identify component that is responsible for sustaining combustion or causing detrimental soot. Accordingly, steps could be taken to cut down the undesirable reaction pathways, claimed Martinez.
He further envisioned that the nanoreactor would help in exploring opportunities for fabricating catalysts that might enhance the efficacy of reactions in applications like fuel cells and other batteries.
The model could also prove helpful in gaining better insights in biochemical reactions that hold important position in human health and drug development.
Source: Stanford University